What Is Imprinting?
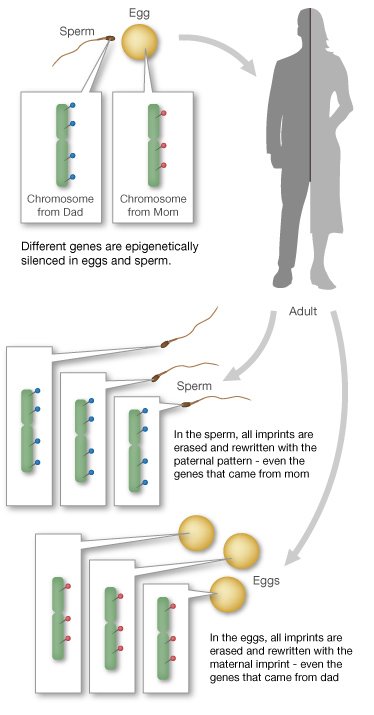
For most genes, we inherit two working copies -- one from mom and one from dad. But with imprinted genes, we inherit only one working copy. Depending on the gene, either the copy from mom or the copy from dad is epigenetically silenced. Silencing usually happens through the addition of methyl groups during egg or sperm formation.
The epigenetic tags on imprinted genes usually stay put for the life of the organism. But they are reset during egg and sperm formation. Regardless of whether they came from mom or dad, certain genes are always silenced in the egg, and others are always silenced in the sperm.