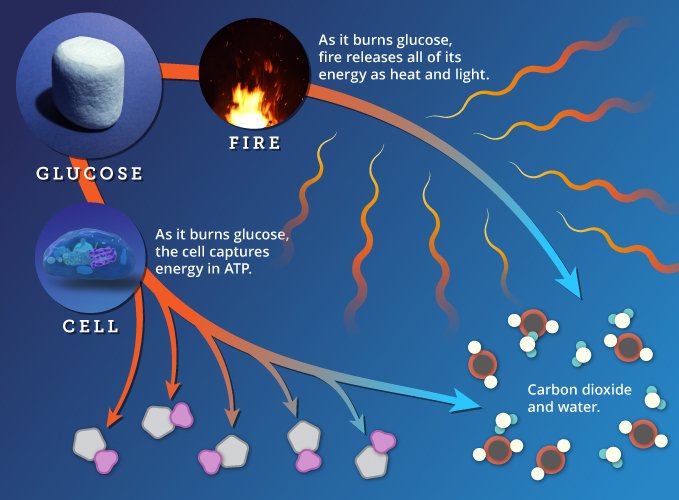
Oxidation and Reduction

In chemical terms, "oxidation" refers the loss of electrons. When we say carbon is oxidized, what we mean is that the carbon atoms in fuel lose electrons as they are converted to carbon dioxide. The electrons they lose are in hydrogen atoms, which are made up of a proton and an electron.
This oxidation reaction is chemically paired with a reduction reaction—what chemists refer to as redox (pronounced REED-ox) reactions. When one atom loses electrons (oxidation), another gains them (reduction). As fuel burns, electrons (in hydrogen atoms) are transferred from carbon to oxygen.
Electrons in glucose (and other fuels) have high "potential energy." In water, they have "fallen" and they have lower potential energy. As atoms are rearranged, the falling electrons release energy. Our cells capture this energy in ATP. An open fire releases it as light and heat.
“LEO says GER” is a simple mnemonic that may help you remember where the electrons go during a redox reaction.
Related content
Visit ATP to learn more about this energetic molecule.