YOUR GOAL: You are studying how a particular gene, named OhNo, might play a role in panic attacks. You want to study mice that are missing this
gene. To "knockout" the OhNo gene, you will replace it with a mutated copy that doesn't work.
Here's how:
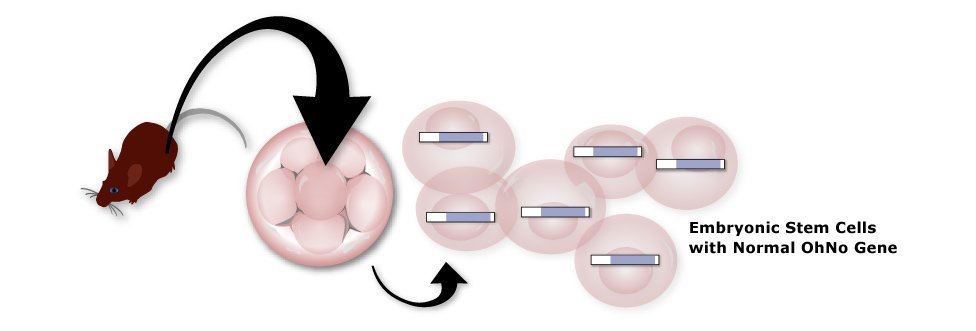
1. Isolate Stem Cells
Isolate embryonic stem cells that originated from male brown mice with a normal OhNo gene (blue).
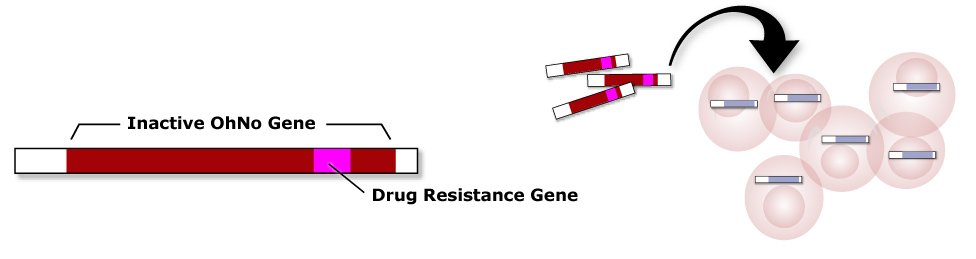
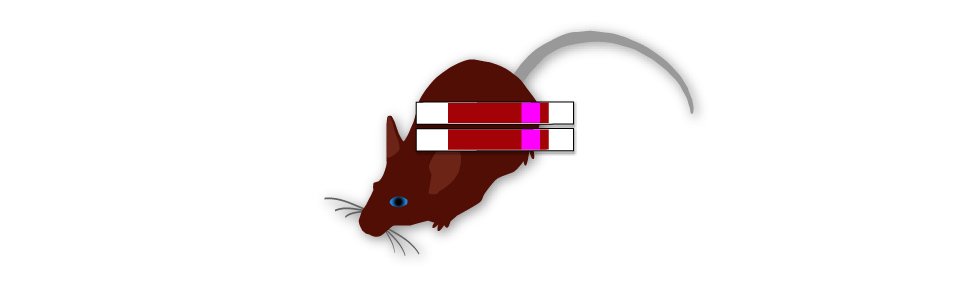
2. Add Inactive Gene With Marker
To these cells, add a copy containing a mutated, inactive OhNo gene (red), and a drug resistance marker gene (pink).
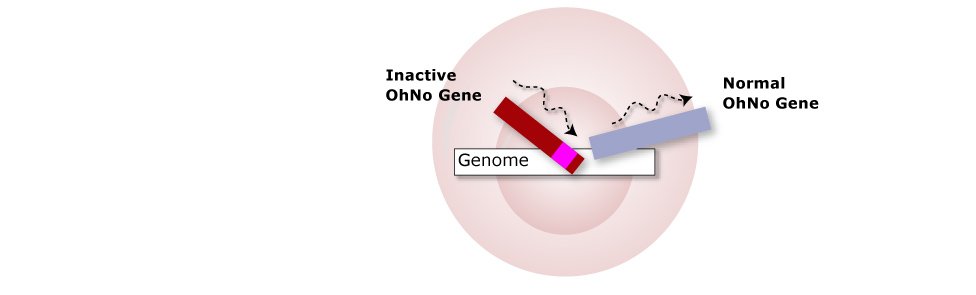
3. Similar Genes Naturally Swap
By mechanisms that are not completely understood yet, similar genes will swap places. The
OhNo gene plus drug resistance marker gene is incorporated into the genome, and the normal version
is kicked out. This process is called homologous recombination.
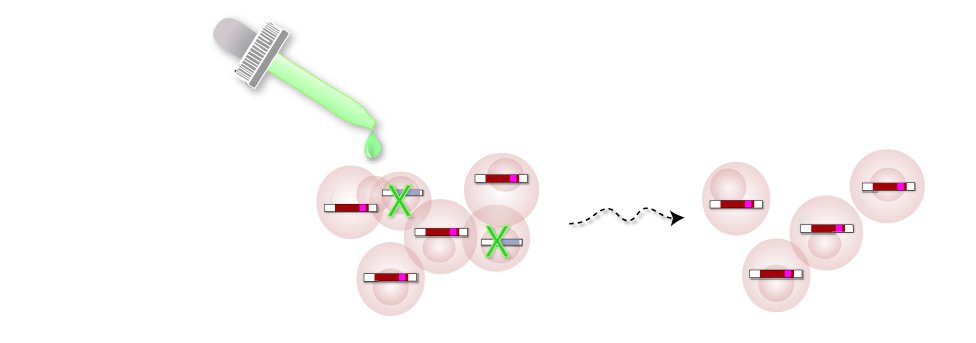
4. Add Drug
Cells that haven't incorporated the inactive OhNo gene don't have the drug resistance marker gene (pink).
Adding the drug kills cells without the marker, leaving you with only cells that have an inactive version of the OhNo gene.
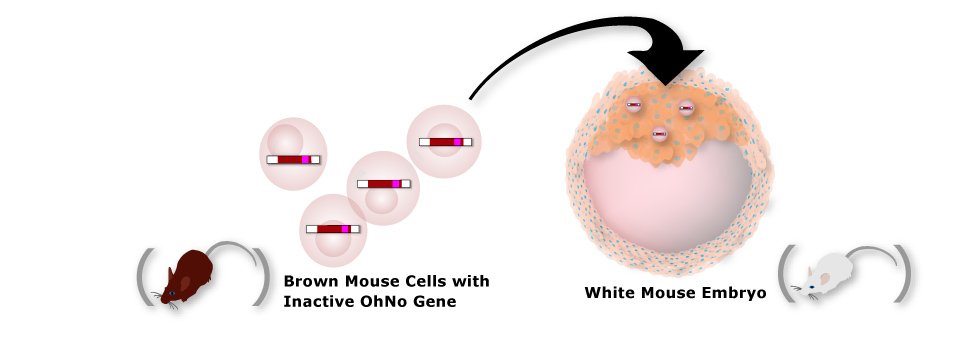
5. Grow Chimeric Mice
By transplanting stem cells that carry the inactive ohNo gene into a white mouse embyro, you'll create what is called a chimera.
Chimeras have patches of cells throughout their bodies that grew from white mouse cells and patches that grew from brown stem cells.
Some of the cells that have the inactive OhNo gene may develop into reproductive cells.
Chimeras are easy to identify because they have both brown and white patches of fur.
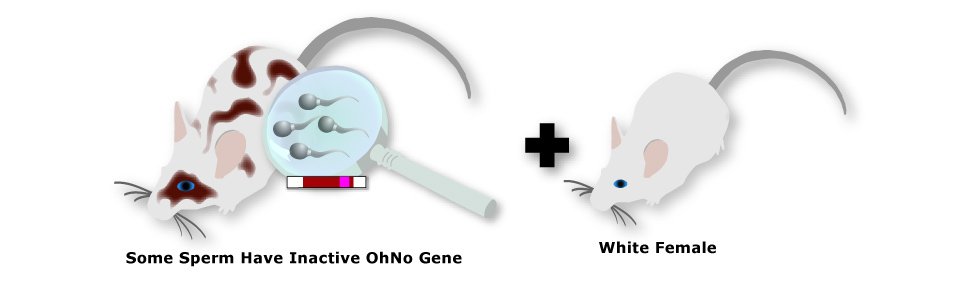
6. Mate Male Chimera
If a male chimera has some reproductive cells (sperm) that originated from the brown stem cells, he will produce
some brown offspring when mated with a white female.
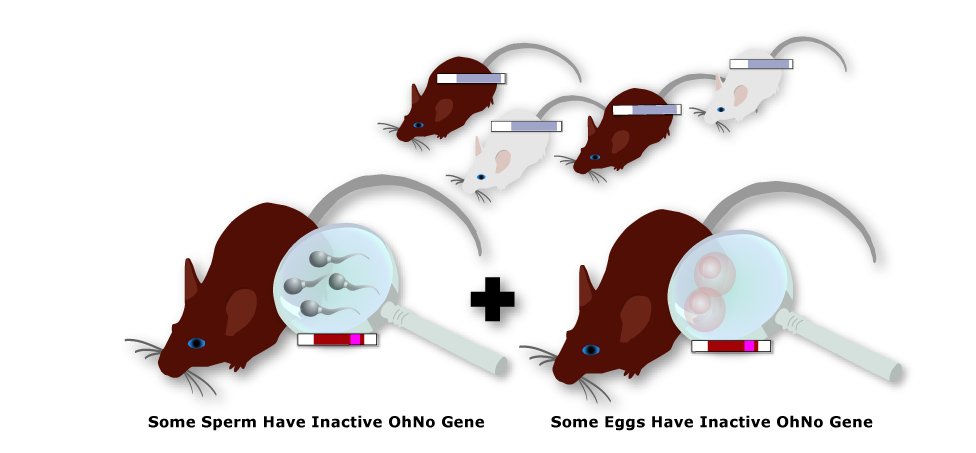
7. Test and Breed Brown Offspring
Half of the brown offspring will have a copy of the inactive OhNo gene in all of their cells—including
their reproductive cells. These mice have one normal copy of the OhNo gene from their mother (not shown) and one inactive
copy from their father. So half of their reproductive cells will contain a normal copy, and half will contain an inactive copy.
These mice can be identified by performing DNA sequencing in their OhNo genes and then bred with each other.
8. You've Made a Knockout Mouse
One fourth of your resulting offsping will have two copies of the "knocked-out" or inactive OhNo gene. You can now
study these mice to determine how lacking the OhNo gene may affect panic attacks.