What is Mutation?
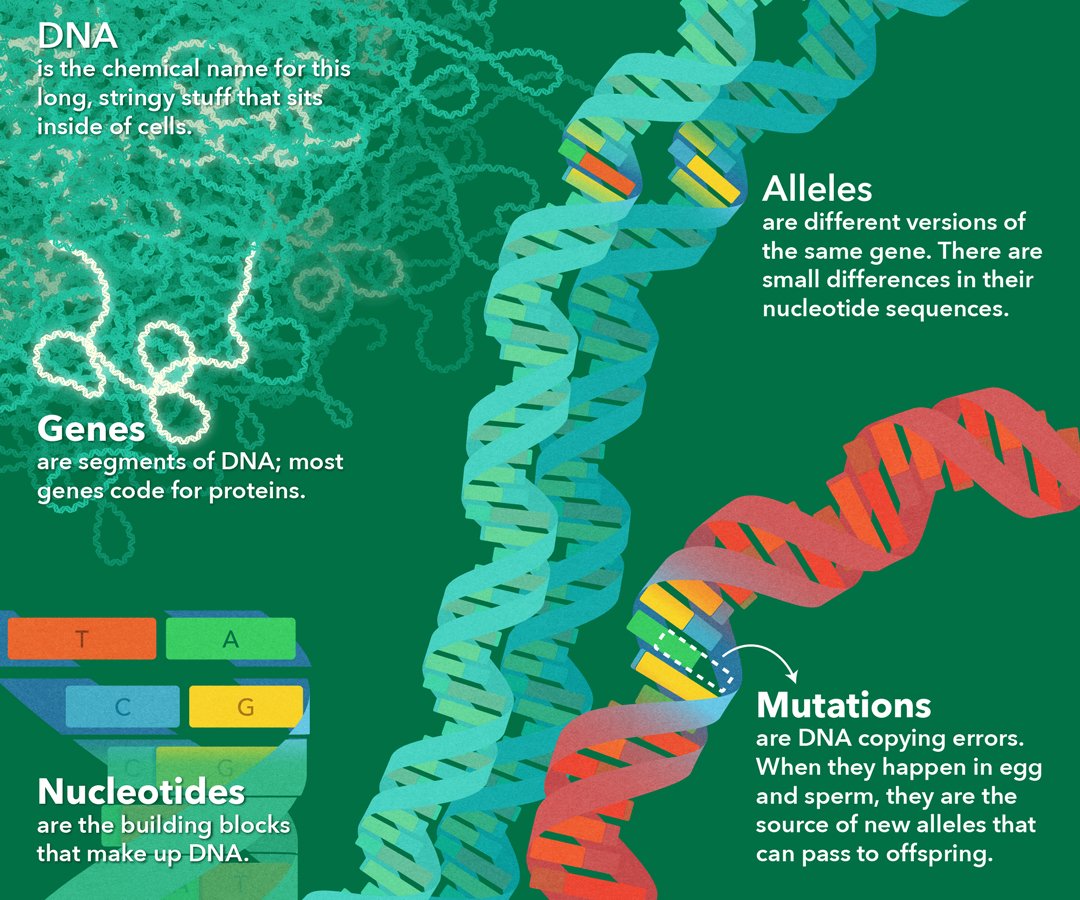
Mutation Generates New Alleles
The whole human family is one species with the same genes. Mutation creates slightly different versions of the same genes, called alleles. These small differences in DNA sequence make every individual unique. They account for the variation we see in human hair color, skin color, height, shape, behavior, and susceptibility to disease. Individuals in other species vary too, in both physical appearance and behavior.
Genetic variation is useful because it helps populations change over time. Variations that help an organism survive and reproduce are passed on to the next generation. Variations that hinder survival and reproduction are eliminated from the population. This process of natural selection can lead to significant changes in the appearance, behavior, or physiology of individuals in a population, in just a few generations.
Once new alleles arise, meiosis and sexual reproduction combine different alleles in new ways to increase genetic variation.

Mutation vs. variation
It's useful to think of mutation as a process that creates genetic variation. We often refer to a mutation as a thing—the genetic variation itself. This approach can be useful when it comes to a gene associated with a disease: the disease allele carries a mutation, a DNA change that compromises the protein's function. However, this approach gives mutation a bad name.
It’s important to remember that losing the function of a gene doesn’t always affect health. For example, most mammals have hundreds of genes that code for olfactory receptors, proteins that help us smell. Losing one of these genes probably doesn’t make all that much difference.
In contrast to variations that cause disease, there are many more examples of variations that are neither good nor bad, but just different—like blood types and eye color. Just like with disease alleles, the process of mutation creates these more neutral variations. But with neutral variations, it can be impossible to tell which allele is the "normal" one that existed first and which is the "mutant"—and the distinction is often meaningless.

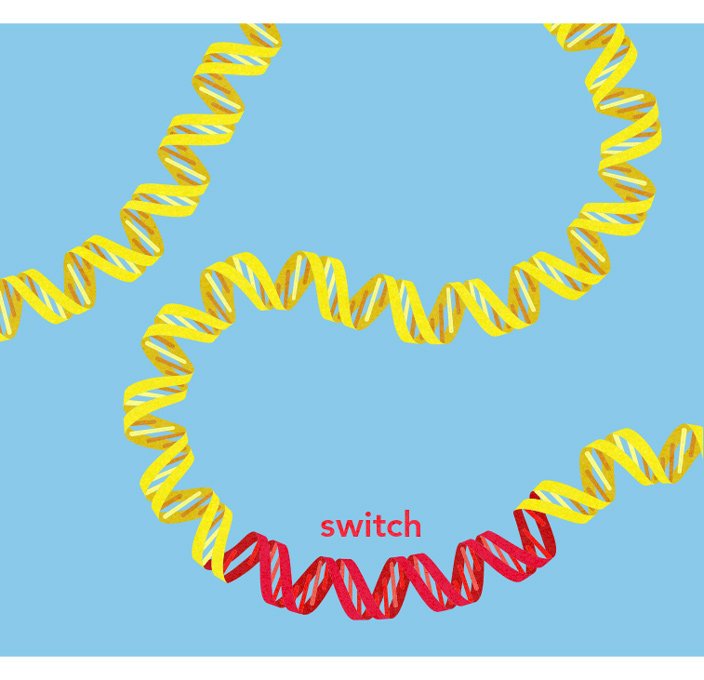
Proteins and switches
Mutation creates variations in protein-coding portions of genes that can affect the protein itself. But even more often, it creates variations in the "switches" that control when and where a protein is active and how much protein is made.
Lactase is an enzyme that helps infants break down lactose, a sugar in milk. Normally the gene that codes for lactase is active in babies and then turned off at about age four. When people who don't make lactase consume milk, they experience gas, nausea, and discomfort. But some people have a variation in a genetic switch that keeps the lactase gene active. This variation is called "lactase persistence," and people who have it can keep milk in their diets even as adults.
Other drivers of mutation: Environmental agents
Radiation, chemicals, byproducts of cellular metabolism, free radicals, ultraviolet rays from the sun—these agents damage thousands of nucleotides in each of our cells every day. They affect the nucleotides themselves: converting one base to another, knocking a base off its backbone, or even causing a break in the DNA strand.

DNA Repair
Most of the time, mutation is reversed. DNA repair machines are constantly at work in our cells, fixing mismatched nucleotides and splicing broken DNA strands back together. Yet some DNA changes remain. If a cell accumulates too many changes—if its DNA is so damaged that repair machinery cannot fix it—it either stops dividing or it self-destructs. If any of these processes go wrong, the cell could become cancerous.
When we put on sun screen, we are protecting ourselves against mutation in somatic cells—the cells that make up the body and are not involved in reproduction. Only when DNA changes are carried in egg and sperm cells are they passed to the next generation. Believe it or not, a certain amount of sloppiness is built into the system. Without mutation there would be no variation, and without variation there would be no evolution.
References
References
Baer, C. F., Miyamoto, M. M., & Denver, D. R. (2007). Mutation rate variation in multicellular eukaryotes: causes and consequences. Nature Reviews Genetics, 8(8), 619-631.
Barnes, D. E., & Lindahl, T. (2004). Repair and genetic consequences of endogenous DNA base damage in mammalian cells. Annu. Rev. Genet., 38, 445-476.
Campbell, C. D., & Eichler, E. E. (2013). Properties and rates of germline mutations in humans. Trends in Genetics, 29(10), 575-584.
Hoeijmakers, J. H. (2009). DNA damage, aging, and cancer. New England Journal of Medicine, 361(15), 1475-1485.
Jackson, S. P., & Bartek, J. (2009). The DNA-damage response in human biology and disease. Nature, 461(7267), 1071-1078.
Roach, J. C., Glusman, G., Smit, A. F., Huff, C. D., Hubley, R., Shannon, P. T., ... & Shendure, J. (2010). Analysis of genetic inheritance in a family quartet by whole-genome sequencing. Science, 328(5978), 636-639. doi:10.1126/science.1186802